
MILSAPERIDONE
Table of Contents
MILSAPERIDONE :
Product Description:
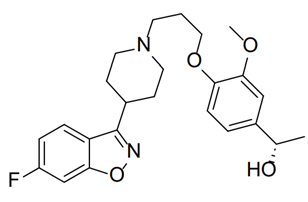
Milsaperidone is an investigational atypical antipsychotic belonging to the piperidinyl-benzisoxazole derivative chemical class. It is structurally related to several second-generation antipsychotic agents and has been designed to provide effective control of psychotic symptoms while minimizing adverse effects commonly associated with older antipsychotic medications.
Chemically, Milsaperidone appears as a white to off-white, finely crystalline powder with moderate lipophilicity, allowing adequate penetration across the blood–brain barrier. The compound is typically formulated for oral administration, although other dosage forms may be explored during development. Its physicochemical properties, including crystalline nature and stability under controlled conditions, support its suitability for pharmaceutical formulation and large-scale manufacturing.
Pharmacologically, Milsaperidone acts primarily through dopamine D₂ receptor antagonism and serotonin 5-HT₂A receptor modulation, mechanisms commonly associated with atypical antipsychotics. This dual receptor activity helps balance dopaminergic and serotonergic neurotransmission in the central nervous system, which plays a key role in reducing symptoms such as hallucinations, delusions, and disorganized thinking. Because of this receptor profile, the drug may also exhibit a lower risk of extrapyramidal side effects compared with first-generation antipsychotics.
Therapeutically, Milsaperidone is being investigated for the treatment of Schizophrenia and other psychotic disorders. Early pharmacological studies suggest that it may provide antipsychotic efficacy with improved tolerability, particularly regarding metabolic effects and movement-related adverse events. Ongoing clinical research aims to evaluate its safety, optimal dosing regimen, and long-term efficacy in patients with schizophrenia and related psychiatric conditions.
Overall, Milsaperidone represents a promising candidate within the class of second-generation antipsychotic agents, with potential advantages in efficacy, safety, and patient compliance compared to existing therapies.
- Brand name: BYSANTI
- Developer: VANDA PHARMACEUTICALS INC
- Drug class: C‑type natriuretic peptide
- Therapeutic area: schizophrenia
(1S)-1-[4-[3-[4-(6-fluoro-1,2-benzoxazol-3-yl)piperidin-1-yl]propoxy]-3-methoxyphenyl]ethanol
(1S)-1-[4-[3-[4-(6-fluoro-1,2-benzoxazol-3-yl)piperidin-1-yl]propoxy]-3-methoxyphenyl]ethanol
(S)-Hydroxy Iloperidone
(|AS)-4-[3-[4-(6-Fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3-methoxy-|A-methylbenzenemethanol
(alphaS)-4-[3-[4-(6-Fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3-methoxy-alpha-methylbenzenemethanol
(S)-1-(4-(3-(4-(6-fluorobenzo[d]isoxazol-3-yl)piperidin-1-yl)propoxy)-3-methoxyphenyl)ethan-1-ol
(S)-1-[4-{3-[4-(6-fluoro-benzo(d)isoxazol-3-yl)-piperidin-1-yl]-propoxy}-3-methoxv-phenyl]-ethanol
(S)-1-[4-{3-[4-(6-fluoro-benzo(d)isoxazol-3-yl)-piperidin-1-yl]-propoxy}-3-methoxy-phenyl]ethanol
OB Approval Data:
| Brand name | : | BYSANTI |
| Developer | VANDA PHARMACEUTICALS INC | |
| Molecular formula | : | C24H29FN2O4 |
| Molecular weight | : | 428.50 g/mol |
| IUPAC Name | : | Benzenemethanol, 4-[3-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1- piperidinyl]propoxy]-3-methoxy-α- methyl-, (αS)- |
| CAS number | : | 501373-88-2 |
| Strength | : | 1, 2, 4, 6, 8, 10 & 12 MG |
| Dosage form/Route | : | Tablet/Oral |
| Approval date | : | Feb 20, 2026 |
| Mechanism of Action | : | The mechanism of action of milsaperidone in the treatment of schizophrenia in adults and the acute treatment of manic or mixed episodes associated with bipolar I disorder in adults is unknown. However, the efficacy of milsaperidone in these conditions could be mediated through a combination of dopamine type 2 (D2) and serotonin type 2 (5-HT2) antagonism. Milsaperidone and iloperidone rapidly interconvert in vivo. Milsaperidone has an in vitro receptor binding profile similar to iloperidone. |
| Maximum daily dosage | : | 24 mg |
| Proposed Indication | : | Treatment of schizophrenia in adults |
| Inactive ingredients (Excipients) | : | olloidal silicon dioxide, crospovidone, hydroxypropylmethylcellulose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and water (removed during processing). |
| Storage Temperature/ Conditions | : | Store the tablets at controlled room temperature, 20°C to 25°C (68°F to 77°F), with excursions permitted between 15°C to 30 °C (59°F to 86°F) |
Clinical Trial Information for Milsaperidone
Overview
Milsaperidone is an atypical antipsychotic developed by Vanda Pharmaceuticals for the treatment of psychiatric disorders such as Schizophrenia and Bipolar I Disorder. The drug has been evaluated in several randomized, placebo-controlled clinical trials to determine its efficacy, safety, and optimal dosing.
Key Phase III Clinical Trials
ENLIGHTEN-1 Trial
- Phase: Phase III
- Study Design: Randomized, double-blind, placebo-controlled trial
- Population: Adults with acute exacerbation of Schizophrenia
- Intervention: Oral Milsaperidone 75 mg/day vs placebo
- Participants: Part of a combined population of 1,498 patients across pivotal trials
- Primary Endpoint: Change in Positive and Negative Syndrome Scale (PANSS) score
Results:
Treatment with milsaperidone significantly reduced PANSS scores by 17.2 points compared with 7.4 points for placebo at week 4, demonstrating statistically significant improvement in schizophrenia symptoms.
ENLIGHTEN-2 Trial
- Phase: Phase III
- Study Design: Randomized, double-blind, placebo-controlled study
- Population: Adults with schizophrenia
- Intervention: Milsaperidone 100 mg/day vs placebo
Results:
Patients receiving milsaperidone showed a 20.6-point reduction in PANSS scores, compared with 9.1 points in the placebo group, indicating strong antipsychotic efficacy comparable to established agents such as Risperidone.
DAYBREAK-1 Trial
- Phase: Phase III
- Study Design: Randomized, placebo-controlled clinical trial
- Population: Adults experiencing manic or mixed episodes of Bipolar I Disorder
- Participants: 436 patients
Primary Endpoint:
Change in Young Mania Rating Scale (YMRS) score.
Results:
Patients treated with milsaperidone showed a 14.3-point reduction in YMRS scores, compared with 7.8 points for placebo, demonstrating significant improvement in manic symptoms.
Additional Ongoing Clinical Trial
Clinical Trial ID
NCT06830044
- Phase: Phase III
- Study Design: Randomized, double-blind, placebo-controlled multicenter study
- Condition: Major Depressive Disorder
- Participants: Approximately 500 patients aged 18–65
- Intervention: Adjunctive oral Milsaperidone vs placebo
- Study Start: March 2025
- Estimated Completion: March 2028
- Primary Objective: Evaluate efficacy and safety of milsaperidone as an add-on therapy in patients with inadequate response to antidepressants.
Regulatory Outcome
Based on results from these clinical studies, Milsaperidone (marketed as Bysanti) received U.S. FDA approval in 2026 for the treatment of Schizophrenia and acute manic or mixed episodes associated with Bipolar I Disorder in adults.
Synthetic routes:
Patent Literature:
Example 1
(S)-1-(4-(3-r4-(6-Fluoro-benzofd1isoxazol-3-vπ-piperidin-1-vπ-propoxy)-3-methoxy-phenvπ-ethanol
56.36 g of boran complex of (3aR, 7R)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1 ,3,2]oxazaborole (1 equivalent) is dissolved under nitrogen in methylenchloride, and the solution is cooled to 0°C. A 1M solution of 1-(4-{3-[4-(6-fluoro-benzo[d]isoxazol-3-yl)-piperidin-1-yl]-propoxy}-3-methoxy-phenyl)-ethanone (iloperidone; 1 equivalent) in methylenchloride is added via a dropping funnel over 90 minutes while the internal temperature is maintained at 0°C ± 2°C. After the addition is complete, the mixture is stirred at 0°C for 20 hours. The reaction mixture is then poured into precooled methanol (0-5°C) during 1 hour. The solution is warmed to room temperature and stirred until the H2 evolution ceases. The solution is concentrated by distillation and the residue dried in vacuum, treated with methanol and stirred for about 1 hour at 50°C and an additional hour at 0CC. The product is isolated by filtration and dried under reduced pressure for 3 hours at 50°C. The title compound is obtained (white crystals).
[α]D20– 19.3° (c=1 in chloroform)
Mp: 138.2 – 138.8°C
The boran complex used as starting material can be obtained as follows:
200 ml of a solution of (3aR, 7R)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole (1M in toluene) is stirred at room temperature under nitrogen. 1.2 equivalent borane-dimethylsulfide complex is added with a syringe. The solution is stirred for 2 further hours at room temperature. The borane complex is then crystallised by addition of 4 vol dry hexane and cooling to -12°C for 1.5 hour. The product is isolated by filtration in a sintered glass funnel and dried in vacuum at 40°C. The boran complex is obtained /white crystals).
Example 2
(R)-1-(4-(3-r4-(6-Fluoro-benzord1isoxazol-3-vπ-piperidin-1-vn-propoxy)-3-methoxy-phenlvπ-ethanol
This compound is produced in analogy to Example 1, using boran complex of (3aS, 7R)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole.
[α]D20 = + 18.4° (c=1 in chloroform)
Mp: 137.9 – 138.3°C
- Follow for more : LatestDrugapproval
